 
Cations: are positively (+) charged ions. To remember this name notice that it has a t in it which looks like. The resulting anion is O 2 − with electron configuration, 1 s 22 s 22 p 6. The difference between cations and anions arises from their different charge. because they have more PROTONS than ELECTRONS. 
The second shell has six electrons ( 2 s 22 p 4) and needs two electrons to achieve octet. Answer (1 of 5): An ion, by definition, is a system (atom or compound) that has a net nonzero electric charge. The electron configuration of O atom is 1 s 22 s 22 p 4. Notable negative ions are chloride and fluoride. 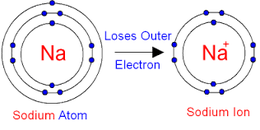
How many electrons must O lose/gain to achieve octet? Write the formula of the resulting ion and its electron configuration. When the electron count is more than the number of protons, the result is a negative ion. Write the electron configuration of oxygen atom (Z=8). The total electric charge of the atom is therefore zero and the atom is said to be neutral. In macroscopic samples of sodium chloride, there are billions and billions of sodium and chloride ions, although there is always the same number of cations and anions. When an atom has an equal number of electrons and protons, it has an equal number of negative electric charges (the electrons) and positive electric charges (the protons). The number of electrons lost by the sodium atom (one) equals the number of electrons gained by the chlorine atom (one), so the compound is electrically neutral. Notice that there are no leftover electrons. Study with Quizlet and memorize flashcards containing terms like A positive ion has more a) electrons than neutrons b) electrons than protons c) protons than neutrons d) protons than electrons e) neutrons than protons, 2 protons attract each other gravitationally and repel each other electrically. The resulting combination is the compound sodium chloride. With two oppositely charged ions, there is an electrostatic attraction between them because opposite charges attract. On the right, the chloride ion has 18 electrons and has a 1− charge. On the left, the chlorine atom has 17 electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
